Powerful bleed protection for people living with hemophilia B and A
Royal lives with hemophilia B with inhibitors, uses Alhemo®, and is an employee of Novo Nordisk.
Alhemo® offers bleed prevention with proven results
Results from a clinical trial with people who have hemophilia B or hemophilia A
Primary study results in people with hemophilia B
Treated spontaneous and traumatic bleeds in people with hemophilia B
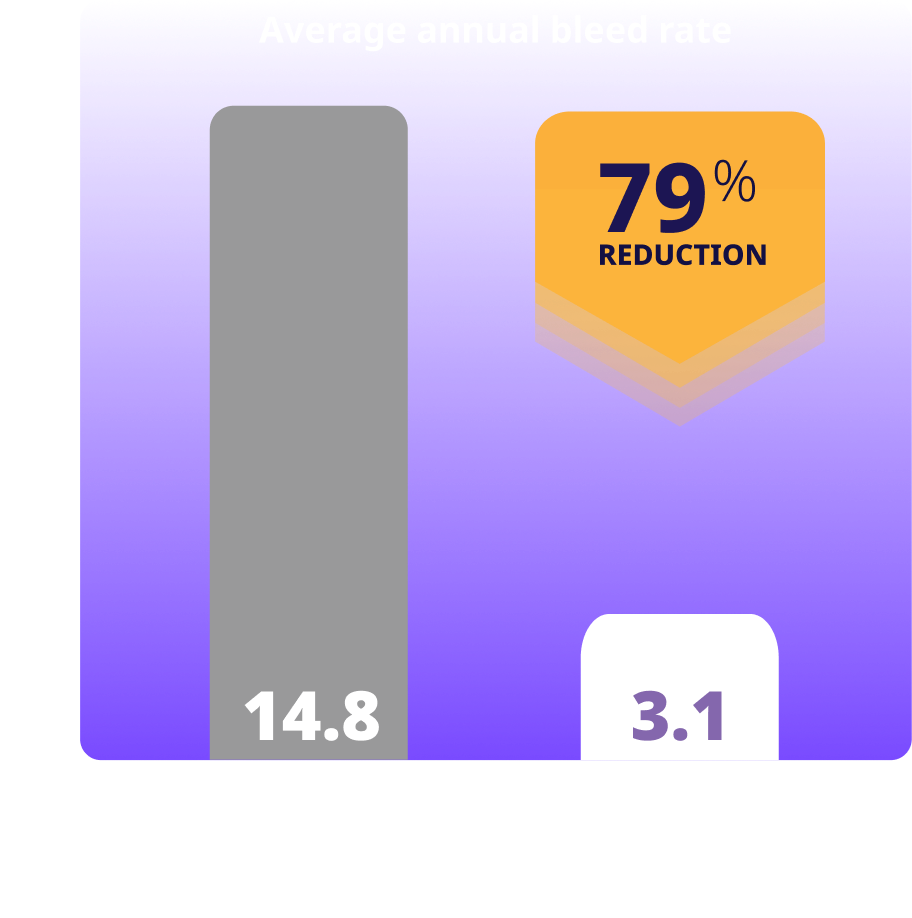
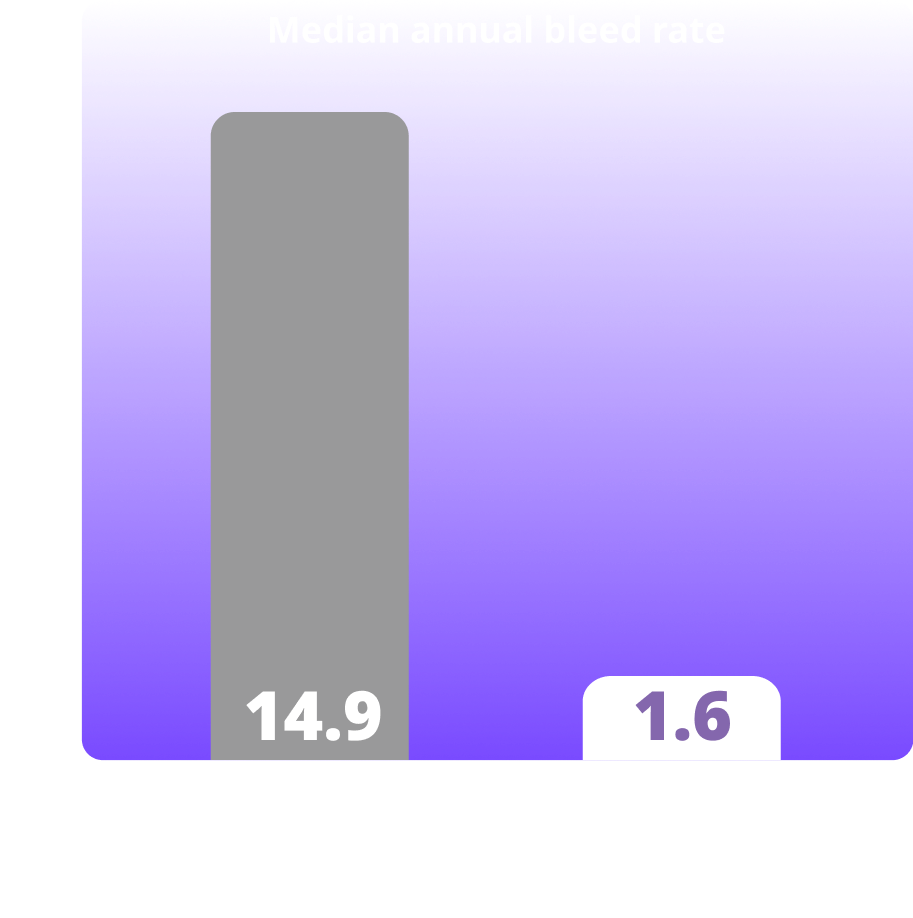
Clinical study of Alhemo® included 156 people aged 12 years and older who had hemophilia A or B without inhibitors. Alhemo® was assessed in a group of people who had previously received on-demand factor replacement. 24 people with HB received Alhemo and 12 people with hemophilia B were treated on-demand with factor 9 replacement. The average ABR was 3.1 for people treated with Alhemo® and 14.8 for people treated with on-demand Factor 9. The median ABR was 1.6 for people on Alhemo® and 14.9 for people treated with on-demand Factor 9. For people with hemophilia A (27 people), the average ABR was 19.3 for those using on-demand Factor 8 and 2.7 for those using Alhemo® prophylaxis. This corresponded to a reduction of 86%.
Royal lives with hemophilia B with inhibitors, uses Alhemo®, and is an employee of Novo Nordisk.
Safety confirmed in multiple clinical trials
people in clinical trials
received at least one dose of Alhemo® prophylaxis, including 66 males with hemophilia B
In the clinical trial with people who have hemophilia B or hemophilia A randomized to receive Alhemo®, injection site reactions and headaches were the most common side effects, each reported in 7% using Alhemo®.
Majority of injection site reactions were mild.
NO boxed warning
NO routine liver monitoring required
Alhemo® is safe to use with all Factor 8 or Factor 9 products. Follow your healthcare provider's instructions on when to use Factor 8 or Factor 9 for breakthrough bleeds, including the recommended dose and schedule.
Royal lives with hemophilia B with inhibitors, uses Alhemo®, and is an employee of Novo Nordisk.
Dosing and administration
Discover personalized dosing and administration steps with Alhemo®.
Michael, Brittany and Martha are Novo Nordisk RBCLs.
Connect with us!
Want to learn more about Alhemo®? Get important email updates, information about patient support programs, and more.


